 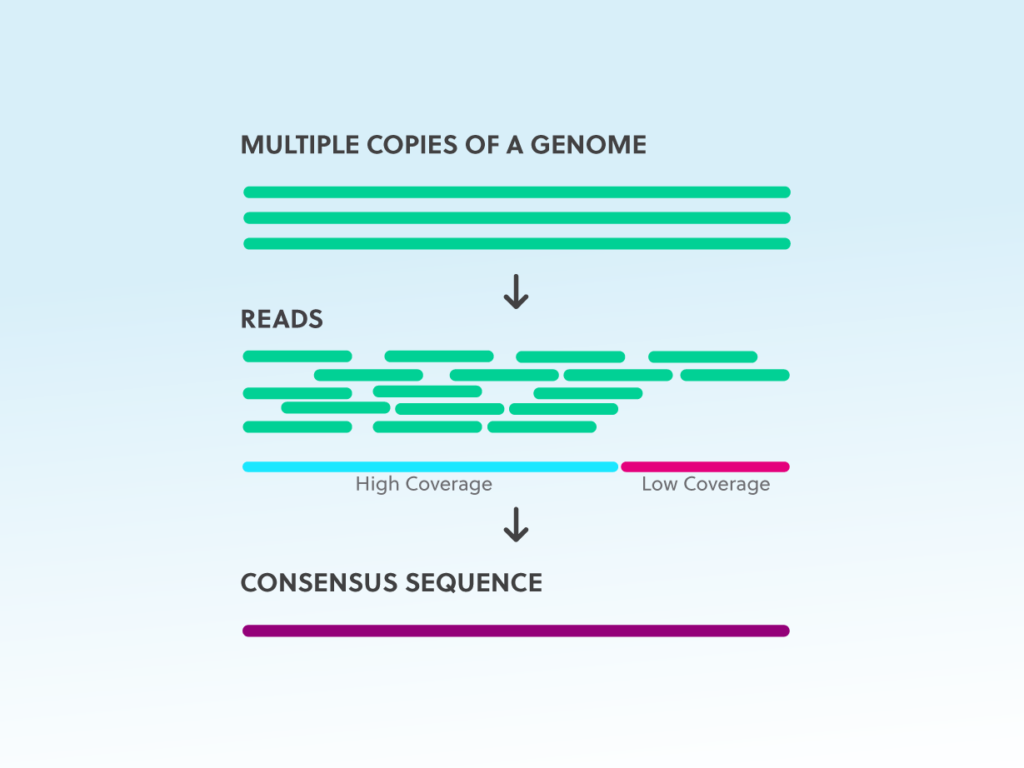
Using the same principle of the methods reported above, microfluidic platforms reliant on individual cell compartmentation in microwells or droplets have been applied in single-cell isolation. Quantitative transcriptomics is used to analyze TCRs and other cell markers, and since each cell is individually barcoded, amplification bias is not an issue. A major improvement in the throughput came with emulsion-based approaches in which single cells are encapsulated in water-in-oil emulsions, where cDNA is synthetized thanks to TCR primers and RT-PCR reagents and then sequenced, maintaining native αβ-chains pairing while sequencing both chains. These ultimate technologies are based, for example, on barcoded gel beads mixed with cells, enzymes and partitioning oil, used to generate V(D)J gene expression libraries (e.g., 10X Genomics, Pleasanton, CA, USA). Researchers have often performed initial bulk analysis and moved, after selection of features of interest, such as binding affinity, to single-cell, even if recently developed commercial single-cell sequencing solutions start to provide full-length paired αβ-chains sequencing of many T-lymphocytes. 
Because of the above-mentioned considerations, single-cell is still a more expensive method compared to other sequencing techniques. Moreover, data analysis requires specific tools and expertise regarding the most appropriate analytical approaches. If starting from cryopreserved cells, it must be tested whether the process of cryopreservation has changed or damaged the cell viability and/or phenotype. Compared with TCR bulk sequencing, the number of cells sequenced using the single-cell approach drops to 10 2–10 3 instead of up to 10 6, and another consideration to take into account is that isolating single cells can be challenging, and obtaining viable cells at the end of the process requires care and to work quickly, with a consequent decrease in the number of samples analyzed and an increase in variability in any single-cell study due to the process workflow. Using mRNA, single-cell TCR sequencing makes it possible to evaluate cell transcriptional heterogeneity down to the single nucleotide level and gene expression variability at the single cell level, leveraging the study of phenotypically different cells populations to an unprecedented resolution. The possibility of extending the TCR repertoire to HLA characterization will be of pivotal importance in the near future to understand how specific HLA genes shape T-cell responses in different pathological contexts and will add a level of comprehension that was unthinkable just a few years ago. Here, an updated overview of TCR repertoire sequencing strategies, providers and applications to infectious diseases and cancer to guide researchers’ choice through the multitude of available options is provided. Although TCR repertoire sequencing is easily accessible today, it is important to deeply understand the available technologies for choosing the best fit for the specific experimental needs and questions. Today, high-throughput sequencing (HTS) applied to the TCR repertoire has opened a window of opportunity to disclose T-cell repertoire development and behavior down to the clonal level. 
T-cells are important players in adaptive immunity and, together with B-cells, define specificity and monitor the internal and external signals that our organism perceives through its specific receptors, TCRs and BCRs, respectively. Many pathological conditions depend on how our immune system responds or does not respond to a pathogen or a disease or on how the regulation of immunity is altered by the disease itself. Indeed, the type and level of exposure to physical and biological agents shape the development and behavior of this complex and diffuse system. The immune system is a dynamic feature of each individual and a footprint of our unique internal and external exposures.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
